Hydroboration

In organic chemistry, hydroboration refers to the addition of a hydrogen-boron bond to certain double and triple bonds involving carbon (C=C, C=N, C=O, and C≡C). This chemical reaction is useful in the organic synthesis of organic compounds.[1]
Hydroboration produces organoborane compounds that react with a variety of reagents to produce useful compounds, such as alcohols, amines, or alkyl halides. The most widely known reaction of the organoboranes is oxidation to produce alcohols from alkenes.
The development of this technology and the underlying concepts were recognized by the Nobel Prize in Chemistry to Herbert C. Brown.[2][3]
Borane adducts
[edit]
Much of the original work on hydroboration employed diborane as a source of BH3. Usually however, borane dimethylsulfide complex BH3S(CH3)2 (BMS) is used instead.[5] It can be obtained in highly concentrated forms.[6]
The adduct BH3(THF) is also commercially available as THF solutions. Its shelf life is less that BMS.[7]
In terms of synthetic results, diborane or the more conveniently handle BMS and borane-THF are equivalent.
Hydroboration of alkenes
[edit]The stoichiometry and idealized regiochemistry of hydroboration of terminal alkenes follows:
- BH3 + 3 RCH=CH2 → B(CH2−CH2R)3
In reality, each hydroboration step follows 1,2-addition but ca. 4% gives the 2,1 addition (affording the B(CH(CH3)R isomer).[1] In extreme cases, such as risubstituted alkenes, hydroboration affords. This significant rate difference in producing di- and tri-alkyl boranes is useful in the synthesis of bulky boranes that can enhance regioselectivity.
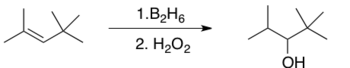
In terms of regiochemistry, hydroboration is typically anti-Markovnikov, i.e. the hydrogen adds to the most substituted carbon of the double bond. That the regiochemistry is reverse of a typical HX addition reflects the polarity of the Bδ+-Hδ− bonds. Hydroboration proceeds via a four-membered transition state: the hydrogen and the boron atoms added on the same face of the double bond. Granted that the mechanism is concerted, the formation of the C-B bond proceeds slightly faster than the formation of the C-H bond. As a result, in the transition state, boron develops a partially negative charge while the more substituted carbon bears a partially positive charge. This partial positive charge is better supported by the more substituted carbon. Formally, the reaction is an example of a group transfer reaction. However, an analysis of the orbitals involved reveals that the reaction is 'pseudopericyclic' and not subject to the Woodward–Hoffmann rules for pericyclic reactivity.


Hydroboration of internal alkenes
[edit]Hydroboration of trisubstituted alkenes places boron on the less substituted carbon.[8]
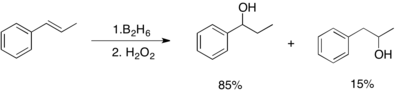
Hydroboration of 1,2-disubstituted alkenes, such as a cis or trans olefin, produces generally a mixture of the two organoboranes of comparable amounts, even if the steric properties of the substituents are very different. For such 1,2-disubstituted olefins, regioselectivity can be observed only when one of the two substituents is a phenyl ring. In such cases, such as trans-1-phenylpropene, the boron atom is placed on the carbon adjacent to the phenyl ring. The observations above indicate that the addition of H-B bond to olefins is under electronic control rather than steric control.
Hydroboration of alkynes
[edit]Hydroboration of alkynes gives alkenylboranes. The stereochemistry is cis-addition. With terminal alkynes, both H2BCH=HR and HB(CH=CHR)2 are formed. Often the hydroboration of alkynes use bulky boranes such as 9-BBN to give monoalkenylborane products. The alkenylboranese are susceptible to many reactions such as protonolysis to give the alkene and oxidation to give the aldehyde or ketone.[9]
Reactions of organoboranes
[edit]As honored by the Nobel Prize to Brown, hydroboration is widely practiced because the alkylboranes are susceptible to many reactions.
Oxidation
[edit]Treatment of alkylboranes with base and hydrogen peroxide gives alcohols:
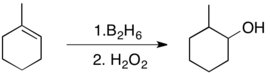
The net reaction is hydration.
Because the addition of H-B to olefins is stereospecific, this oxidation reaction will be diastereoselective when the alkene is trisubstituted.[10] Hydroboration-oxidation is thus an excellent way of producing alcohols in a stereospecific and anti-Markovnikov fashion.
Other C-heteroatom bond forming reactions
[edit]Hydroboration can also lead to amines by treating the intermediate organoboranes with monochloramine or O-hydroxylaminesulfonic acid (HSA).[11]
Terminal olefins are converted to the corresponding alkyl bromides and alkyl iodides by treating the organoborane intermediates with bromine[12] or iodine.[13] Such reactions have not however proven very popular, because succinimide based reagents such as NIS and NBS are more versatile and do not require rigorous conditions as do organoboranes. etc.
Carbonylations
[edit]Trialkylboranes react with carbon monoxide to afford homologated products such as 2-bora-1,3-dioxolanes. When the addition of CO is conducted in the presence of a hydride reducing agent, the primary alcohol is produced.
Specialty boranes for hydroboration
[edit]
One example of a monoalkylborane is thexylborane (ThxBH2), produced by the hydroboration of tetramethylethylene:[14]
- B2H6 + 2 Me2C=CMe2 → [Me2CHCMe2BH2]2
A chiral example is monoisopinocampheylborane. Although often written as IpcBH2, it is a dimer [IpcBH2]2. It is obtained by hydroboration of (−)‐α‐pinene with borane dimethyl sulfide.[15]
Monobromo- and monochloro-borane can be prepared from BMS and the corresponding boron trihalides. The stable complex of monochloroborane and 1,4-dioxane effects hydroboration of terminal alkenes.[16]
Promient among hindered dialkylboranes is disiamylborane, abbreviated Sia2BH. It also is a dimer. Owing to its steric bulk, it selectively hydroborates less hindered, usually terminal alkenes in the presence of more substituted alkenes.[17] Disiamylborane must be freshly prepared as its solutions can only be stored at 0 °C for a few hours. Dicyclohexylborane Chx2BH exhibits improved thermal stability than Sia2BH.
A versatile dialkylborane is 9-BBN. Also called "banana borane", it exists as a dimer. Reactions with 9-BBN typically occur at 60–80 °C, with most alkenes reacting within one hour. Tetrasubstituted alkenes add 9-BBN at elevated temperature. Hydroboration of alkenes with 9-BBN proceeds with excellent regioselectivity. It is more sensitive to steric differences than Sia2BH, perhaps because of it rigid C8 backbone. 9-BBN is more reactive towards alkenes than alkynes.[18]
For catalytic hydroboration, pinacolborane and catecholborane are widely used. They also exhibit higher reactivity toward alkynes.[19] Pinacolborane is also widely used in a catalyst-free hydroborations.
See also
[edit]References
[edit]- ^ a b Herbert C. Brown (1975). Organic Syntheses via Boranes. New York: John Wiley and Sons. ISBN 0471112801.
{{cite book}}: Check|isbn=value: invalid character (help) - ^ Brown, H.C. (1961). "Hydroboration-A Powerful Synthetic Tool". Tetrahedron. 12 (3): 117. doi:10.1016/0040-4020(61)80107-5.
- ^ "The Nobel Prize in Chemistry 1979". www.nobelprize.org. Retrieved 21 March 2017.
- ^ Hutchins, Robert O.; Cistone, Frank (1981). "Utility and Applications of Borane Dimethylsulfide in Organic Synthesis. A Review". Organic Preparations and Procedures International. 13 (3–4): 225. doi:10.1080/00304948109356130.
- ^ See Borane-dimethylsulfide complex
- ^ Zaidlewicz, Marek; Baum, Ofir; Srebnik, Morris (2006). "Borane Dimethyl Sulfide". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rb239.pub2. ISBN 0471936235.
- ^ Kollonitisch, J (1961). "Reductive Ring Cleavage of Tetrahydrofurans by Diborane". J. Am. Chem. Soc. 83 (6): 1515. doi:10.1021/ja01467a056.
- ^ Brown, H. C.; Zwefei, G. (1960). "Isomerization of Organoboranes Derived Addition Mechanism of Isomerization from Branched-Chain and Ring Olefins- Further Evidence for the Elimination-Addition Mechanism of Isomerizaton". Journal of the American Chemical Society. 82: 1504. doi:10.1021/ja01491a058.
- ^ Hudrlik, Paul F.; Hudrlik, Anne M. (1978). "Applications of Acetylenes in Organic Synthesis". In Patai, Saul (ed.). The Carbon–Carbon Triple Bond (1978): Part 1. London: Wiley. doi:10.1002/9780470771563.ch7.
- ^ Allred, E. L.; Sonnenbcrg, J.; Winstcin S. (1960). "Preparation of Homobenzyl and Homoallyl Alcohols by the Hydroboration Method". Journal of Organic Chemistry. 25: 25. doi:10.1021/jo01071a007.
- ^ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, p. 822, ISBN 978-0-471-72091-1
- ^ Brown, H. C.; Lane, C. F. (1970). "The Base-Induced Reaction of Organoboranes with Bromine. A Convenient Procedure for the Anti-Markovnikov Hydrobromination of Terminal Olefins via Hydroboration-Bromination". Journal of the American Chemical Society. 92 (22): 6660. doi:10.1021/ja00725a057.
- ^ Brown, H. C.; Rathke, M.; Rogic, M. M. (1968). "A Fast Reaction of Organoboranes with Iodine under the Influence of Base. A Convenient Procedure for the Conversion of Terminal Olefins into Primary Iodides via Hydroboration-Iodination". Journal of the American Chemical Society. 90 (18): 5038. doi:10.1021/ja01020a056.
- ^ Negishi, Ei-Ichi; Brown, Herbert C. (1974). "Thexylborane-A Highly Versatile Reagent for Organic Synthesis via Hydroboration". Synthesis. 1974 (2): 77–89. doi:10.1055/s-1974-23248.
- ^ Dhar, Raj K.; Josyula, Kanth V. B.; Todd, Robert; Gagare, Pravin D.; Ramachandran, Veeraraghavan (2001). "Diisopinocampheylborane". Encyclopedia of Reagents for Organic Synthesis. pp. 1–10. doi:10.1002/047084289X.rd248.pub3. ISBN 9780470842898.
- ^ Kanth, J. V. B.; Brown, H.C. (2001). "Hydroboration. 97. Synthesis of New Exceptional Chloroborane−Lewis Base Adducts for Hydroboration. Dioxane−Monochloroborane as a Superior Reagent for the Selective Hydroboration of Terminal Alkenes". Journal of Organic Chemistry. 66 (16): 5359–65. doi:10.1021/jo015527o. PMID 11485456.
- ^ Dodd, D.S.; Ochlschlager, A. C. (1992). "Synthesis of inhibitors of 2,3-oxidosqualene-lanosterol cyclase: conjugate addition of organocuprates to N-(carbobenzyloxy)-3-carbomethoxy-5,6-dihydro-4-pyridone". Journal of Organic Chemistry. 57 (10): 2794. doi:10.1021/jo00036a008.
- ^ Dhillon, R. S. (2007). Hydroboration and Organic Synthesis : 9-Borabicyclo [3.3.1] Nonane (9-BBN). Springer.
- ^ Brown, H.C.; Zaidlewicz, M. (2001). Organic Syntheses Via Boranes, Vol. 2. Milwaukee, WI: Aldrich Chemical Co. ISBN 978-0-9708441-0-1.
